Readable in the Dark
the folder of things that don't fit
The Sideline
She was making her own sunscreen.
Not buying it — formulating it, from scratch, mineral-based, because she’d done enough research to have opinions about particle size and UV coverage and the difference between a physical blocker and a chemical one. She knew I was the curator of minerals at the Natural History Museum, so when our kids’ soccer team took the field and we ended up standing next to each other on the sideline, the conversation went somewhere most sideline conversations don’t.
She showed me the bottle. I borrowed some — it was a warm day — and we talked about TiO₂ while the game ran. I told her everything I knew. I didn’t tell her everything I suspected.
I told her what I knew. Titanium dioxide comes in three mineral forms: rutile, anatase, and brookite — chemically identical, architecturally different, each with its own relationship to light and reactivity. Industry chose rutile for sunscreen deliberately. It’s the stable polymorph, the calm one, inert enough to put in food and cosmetics and paper without concern. Rutile powder is one of the whitest materials known — the same mineral that grows as deep red or black crystals in a collection becomes brilliant white when ground small enough. That’s what goes in the bottle.
She asked about the photocatalytic forms — she’d read about anatase generating reactive oxygen species under UV. I told her yes, that’s real, anatase is more active, which is why rutile is preferred for sunscreen. You can eat rutile, spread it on your skin, and it does nothing, which is exactly the point.
We talked for a while longer. The game ran.
The Mineral
Rutile rewards attention precisely because it refuses to look the same twice.



Three specimens, one mineral. The gold twin on hematite. The large silvery-black prismatic crystal from Georgia. The deep red transparent crystals from Minas Gerais. Same chemistry — TiO₂, titanium and oxygen, nothing else — but the color shifts from gold to silver to red depending on trace impurities, crystal size, and the geometry of how light moves through the structure. Collectors love it because it never looks finished. It has the quality of still becoming something.
Ground to powder, it becomes something else entirely: brilliant white. Rutile powder is one of the whitest materials known. It’s in white paint, white paper, white food coating. The bright white of a hospital corridor, the bright white of a wedding cake, the bright white of a printer page — much of that whiteness is rutile. The same mineral, just smaller, with the same crystal structure but a completely different relationship to light.
This is one of the stranger things about minerals at the nanoscale: size changes everything. At 200 nanometers versus 20 nanometers, the same material scatters light differently. Surface area explodes. The mineral doesn’t transform — its chemistry stays identical — but what it does in the world changes entirely.
Industry understood this. That’s why rutile is in sunscreen, in food, in the white of this page. It’s chemically inert across all of those applications. You can eat it. You can spread it on your skin. It does nothing, which is exactly the point.
There is, however, one form of titanium oxide that does something. Something quite different.
That’s the one that had been turning up in the folder.
The Wrong Spectrum
The Peru samples arrived as filters.
A team of climbers and atmospheric scientists had spent weeks in the Cordillera Blanca collecting snow at altitude — physically difficult, logistically complicated work conducted far from any laboratory. What arrived for analysis was the result: membrane filters loaded with particles melted out of high-altitude snow, labeled by site and elevation, ready for mineralogical characterization. The team was looking for black carbon and light-absorbing aerosols. The question was what minerals were present and in what proportions.


The answer, overwhelmingly, was clay. Fine-grained silicate dust carried up from the valleys and páramo below — the expected mineral background of high-altitude Andean snow. Black carbon was present too, the soot signature of combustion at lower elevations transported upward on rising air. Expected things, in expected proportions.
And then, in specific layers, something almost right.
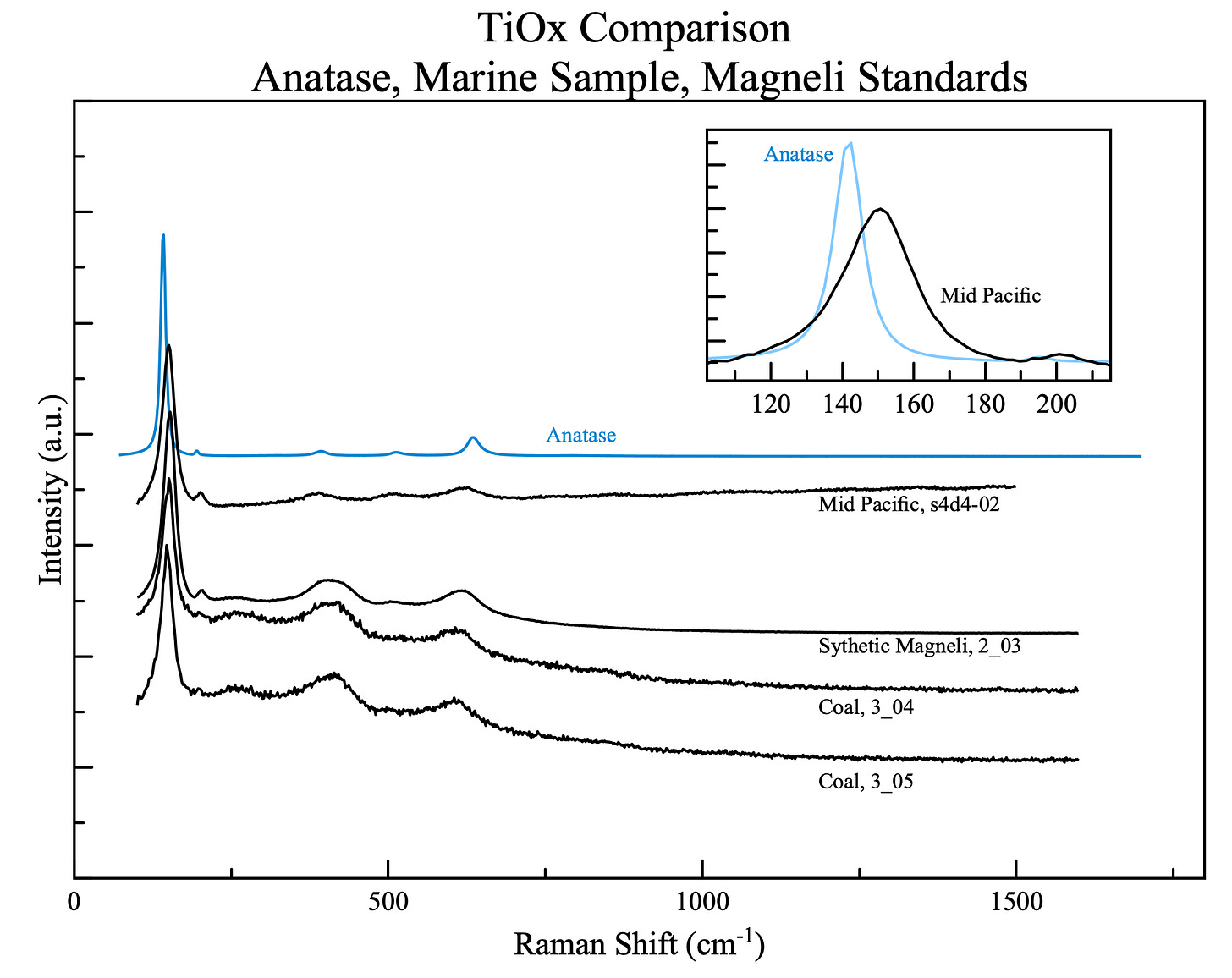
Mineral identification in this kind of work is done with a Raman spectrometer — an instrument that fires a laser at a sample and reads the way the mineral scatters the light back. Every mineral has a characteristic fingerprint: a set of peaks at specific positions, like a barcode. Match the barcode to a reference database, and you have an identification. The instrument is fast, non-destructive, and works on particles too small to see with the naked eye. It is also unforgiving of anything that doesn’t match a reference — if the barcode is slightly wrong, you’re left with a closest match and a note that something is off.
Certain layers contained something in the TiO₂ region of the spectrum — peaks in positions that suggested titanium oxide, in a part of the Raman fingerprint where anatase and brookite live. But the peaks didn’t match either. They didn’t match anything. Not anatase. Not brookite. Not rutile. Not any mineral in the reference database. The barcode didn’t scan wrong — it didn’t scan at all.
The closest match was anatase, so that’s what went into the notes. Not because it was right, but because the database had to return something and “unidentified TiO₂-like phase” is not a mineral name. The data the team had asked for was returned. They got what they needed. The unidentified signal went into a folder, labeled with the closest approximation available, which happened to be wrong.
This is what responsible science actually looks like. You don’t invent explanations for signals that don’t yet have enough context to support one. You document, you flag, you move on.
The folder sat there. The work moved on.
The Same Signal, Everywhere
Except the signal didn’t stay in the folder. It followed.
The next samples came from the Great Salt Lake — brine delivered by a colleague running halite crystallization experiments, asking whether bacteria could survive entombment inside salt crystals as they formed. The Raman work was to identify mineral phases in the brine. The minerals were found and identified. But there it was again: the same shifted TiO₂ signal, persistent across multiple samples, in water from a landlocked basin in the middle of Utah. Labeled anatase-like, noted as anomalous, and the halite mineralogy was delivered as requested.
The Pacific Ocean filters came from Will Berelson’s group at USC and Jess Adkin’s group at Caltech — a North Pacific transect from Hawaii to Alaska, studying how pteropod shells made of aragonite dissolve in the water column and what that means for the ocean’s carbon budget. The Raman contribution was to confirm the carbonate mineralogy on filters collected by in-situ pumps at depth: map the distribution of calcite and aragonite across the filter surfaces, verify what the X-ray diffraction had shown. That work was clean. The carbonate identifications were unambiguous. But the filters also carried non-carbonate particles, and among them — in samples from multiple depths, multiple stations, hundreds of meters below the surface of the open Pacific — the same shifted peaks appeared again. Flagged. Filed. The dissolution study got its carbonate mineralogy.
Above: This is the project the Pacific filters came from. The carbonate mineralogy was clean. The other signal is what this post is about.
The fish gut samples came from Bill Ludt, the ichthyology curator at the museum, who wanted to know whether Raman spectroscopy could identify plastic polymer types in historical fish specimens — a microplastics project using the collection, specimens going back decades. It could. The protocols were worked out. The plastics were there, accumulating through time in the collection just as they accumulate in the ocean. But in open Pacific fish, collected far from any coastline, the gut contents also carried the signal. The same signal. In a fish preserved in ethanol in an archival jar, pulled from a shelf in the collections.
Four separate projects. A glaciologist, a geobiologist, an oceanographer, an ichthyologist. None of them asking the same question, none of them looking for the same thing. Each got the answer they came for. And in the background of every dataset, the same anomalous spectrum — in snow from the Andes, in brine from Utah, in water from the North Pacific, in the gut contents of open-ocean fish.
These places share almost nothing. Different hemispheres. Different altitudes. Different ecosystems. The Andes and the floor of the Pacific Ocean and a landlocked Utah lake and the gut of a fish preserved in a jar — there is no obvious thread connecting them. And yet the same unidentified signal kept appearing in all of them, in samples delivered by people who had never spoken to each other, asking completely different questions.
The connection wasn’t visible yet. But the same material had been called by two different names — sometimes anatase, sometimes something closer to hematite, depending on which peaks dominated in a given sample, depending on which reference mineral happened to be nearest in the database on a given day. Two mineral names. One signal. Four projects. One folder of anomalies that nobody had asked to be explained.
Then, in 2017, a paper arrived.
What It Is
Yang et al., Nature Communications, 2017. A team from Virginia Tech investigating a coal ash spill into a river in North Carolina. They found unexpected particles in the downstream sediment. Almost like rutile. But not. Characteristic superfine striations under electron microscopy. Raman peaks shifted from TiO₂ in a specific, diagnostic way.
Magnéli phases — TiₓO₂ₓ₋₁
The name comes from Arne Magnéli, a Swedish crystallographer who first characterized this family of titanium suboxides in the early 1950s — decades before anyone knew they were being produced by the billions of tons and carried to the top of the world on the wind.
These are titanium suboxides — members of a family of materials formed when TiO₂ is heated above 900°C in a low-oxygen environment. The oxygen atoms are stripped from the rutile structure along specific crystallographic shear planes. What remains is a material with the same titanium backbone but a fundamentally different character. Not white. Black. Not an insulator. A narrow-band semiconductor with electrical conductivity roughly 100,000 times higher than TiO₂. Absorptive across UV, visible, and near-infrared light. A material that, in Parker and Siegel’s 1990 calibration work, produces exactly the kind of shifted, broadened Raman peaks that had been accumulating in that folder for years — peaks that drift between “looks like anatase” and “looks like hematite” depending on which suboxide stoichiometry dominates in a given sample.
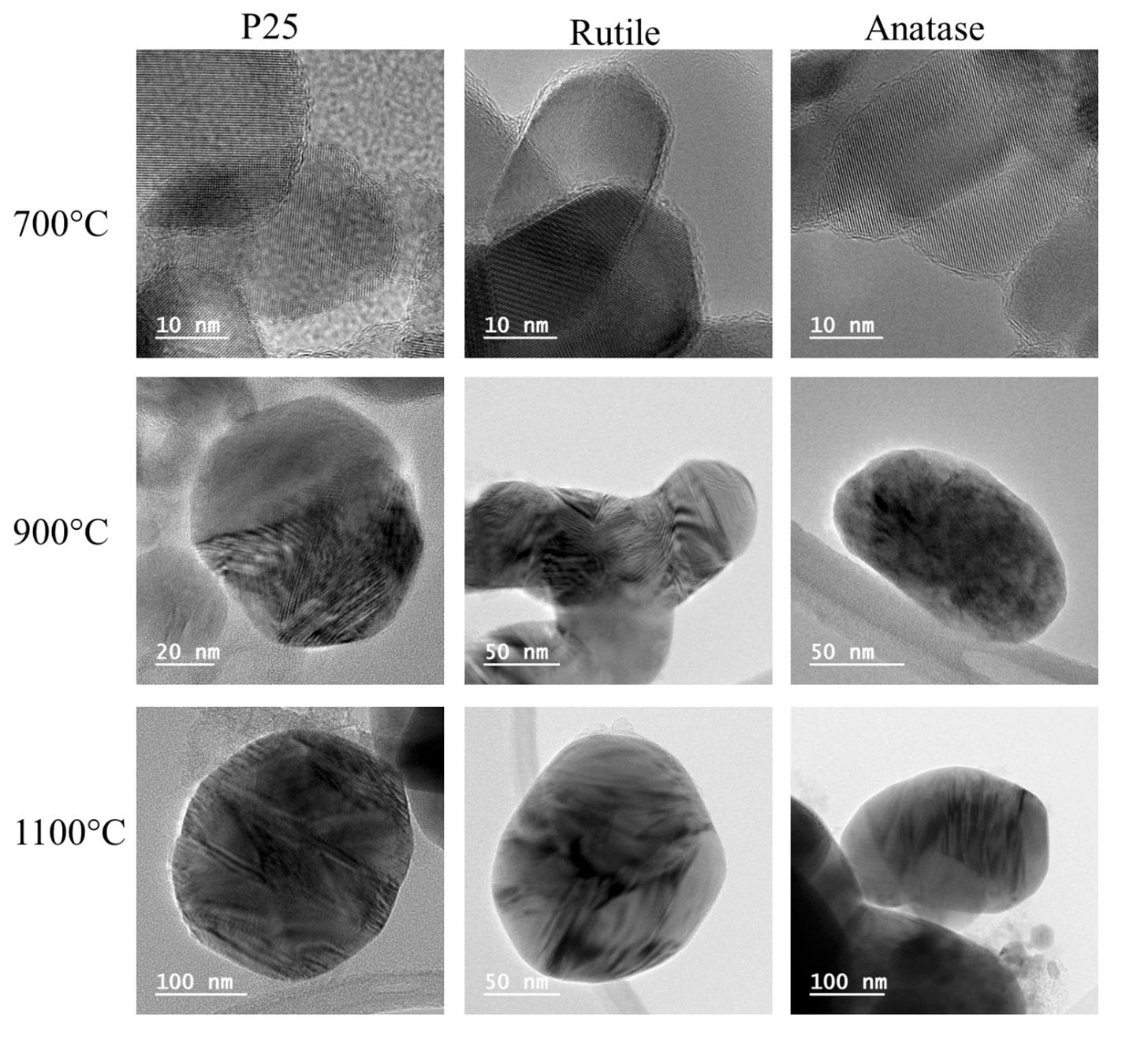
The conditions required to form Magnéli phases — temperatures above 900°C, reducing atmosphere, TiO₂ as a starting material — describe exactly one widely distributed industrial process.
Coal combustion.
TiO₂ is a minor accessory mineral in coal worldwide. It sits there unremarkably in the rock for millions of years. Then it goes into a furnace at 1,400°C with almost no available oxygen, and it comes out the other end as something that has never existed in nature. Coal burning plants in North Carolina. In Kentucky. In Virginia. In Texas. In Shanghai. In Chongqing. Yang et al. tested ash from 22 plants across the United States and China. Every single one contained Magnéli phases.
Every anomalous spectrum in the folder resolved.
The Glacier
Here is where the story stops being abstract.
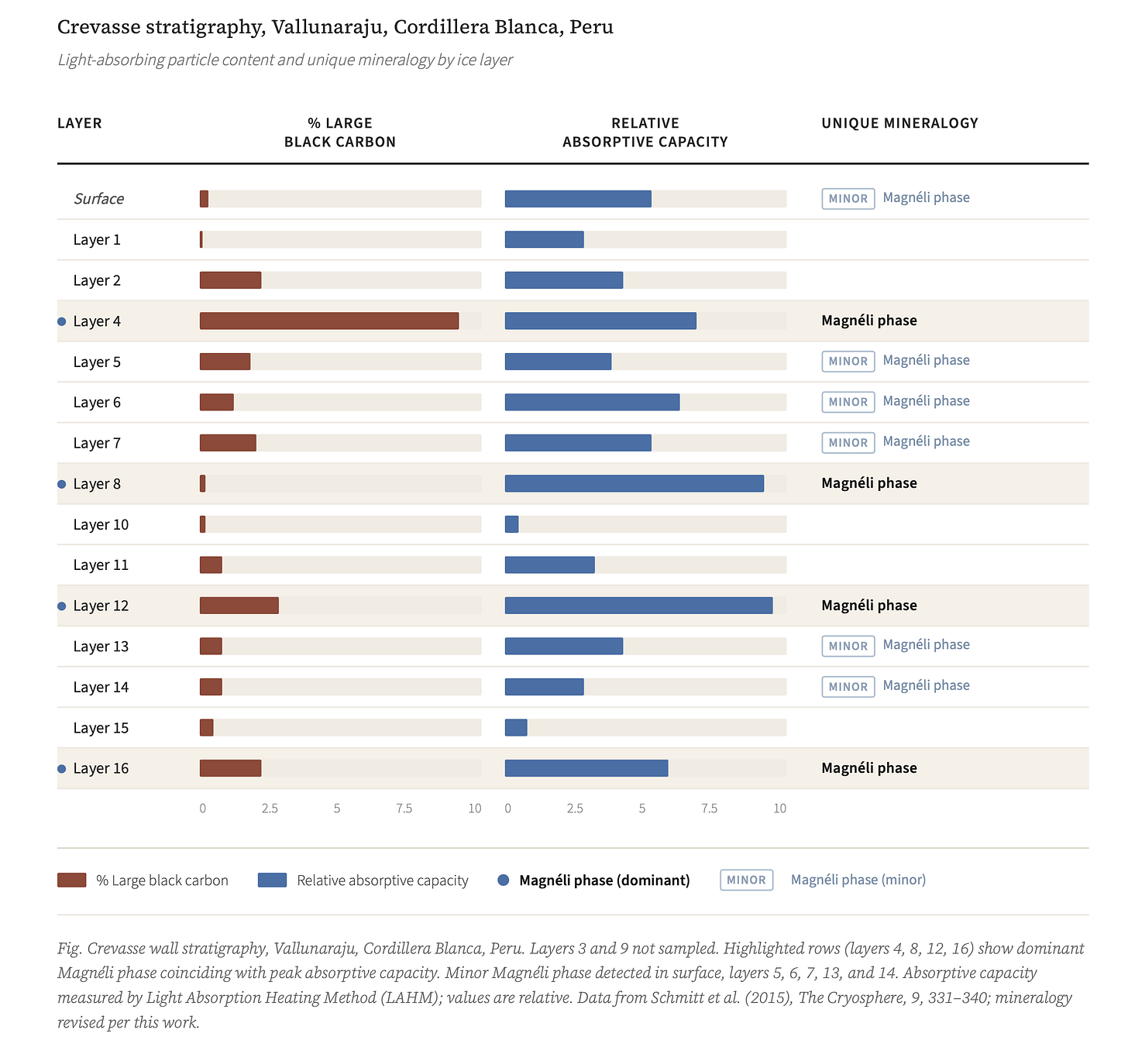
The Peru snow samples came from a specific location: the wall of a crevasse on Vallunaraju mountain — the glaciated peak nearest to Huaraz, the largest city in the region. The crevasse wall showed what glacier walls always show if you look carefully: layers. Dark bands separated by cleaner ice, each band recording a season of accumulation, a year of deposition, a chapter in the mountain’s slow autobiography.
The absorptive capacity of particles in each layer was measured using a technique that exposes filters to visible light and records the temperature increase — the Light Absorption Heating Method. The results showed distinct spikes at layers 4, 8, 12, and 16 in the sequence, with absorptive capacity dramatically higher than the surrounding ice.
The interpretation was black carbon. Coal combustion produces black carbon. Black carbon absorbs light. Black carbon on glaciers accelerates melt. The logic was sound. The data supported it.
Here is what the technique cannot do: distinguish between black carbon and other dark, light-absorbing particles. It measures heat. It was calibrated against black carbon because that was the dominant hypothesis, the reasonable hypothesis. The study was not wrong to reach that conclusion.
Here is what Magnéli phases are: black. Not dark gray, not brown — black, in the way that materials with near-zero band gaps are black, absorbing across nearly the full solar spectrum. Under the LACM technique, a filter loaded with Magnéli phases is indistinguishable from a filter loaded with soot. They produce the same temperature curve. They would be reported as the same effective black carbon concentration.
This is not a theoretical concern about absorption properties. Industry knows exactly what Magnéli phases do with light — they are actively engineered for it. Ti₄O₇, the most common Magnéli phase, is used in photothermal conversion applications specifically because it absorbs solar radiation across the full spectrum and converts it to heat with exceptional efficiency. Engineers are trying to put these materials to work as solar heat absorbers. The same property that makes them commercially attractive is what makes them concerning on the surface of a glacier at 6,000 meters, under some of the most intense UV radiation on Earth.
Coal combustion produces both black carbon and Magnéli phases simultaneously. They travel together in fly ash. They deposit together on glacier surfaces. And in the Raman data from those same Peru samples — the data labeled with the closest available mineral name and set aside — the layers with the highest absorptive capacity are precisely the layers where the unidentified signal appears.
At 6,000 meters, UV flux is among the highest on Earth. Magnéli phases, with their broad-spectrum absorption and their extraordinary conductivity, are photocatalytically active under those conditions in ways that TiO₂ is not. A small concentration of dark, highly absorptive particles on glacier ice, under intense UV, releasing energy directly into the snowpack — the heat capacity calculations are consistent with the observed temperature anomalies.
The Cordillera Blanca has lost 20-30% of its glacier area since 1970. Dry season discharge in some valleys is projected to fall by 70% compared to pre-warming levels. Millions of people depend on glacial meltwater for drinking water and agriculture.
The accelerated melt was attributed to black carbon.
That attribution was not wrong. But it was not complete.
What We Don’t Know
Here is what the data actually shows: Magnéli phases are present wherever Raman spectroscopy has been run on environmental samples from the industrial era — high-altitude glacial ice, the open Pacific Ocean, the gut contents of museum fish, a terminal lake basin in the middle of Utah. Coal combustion is the dominant known mechanism for producing them outside a laboratory, though recent work has identified combustion of TiO₂-rich synthetic materials in urban structure fires as an additional source. They co-occur with the high-absorption layers in the Peru glacier stratigraphy.
That much is solid ground.
What isn’t solid is what any of it means at scale.
The glacier melt question is the one that lingers. The LACM technique — used to measure light absorption in those Peruvian ice cores — cannot distinguish between black carbon and Magnéli phases. Both are dark. Both absorb broadly. Both are combustion products that travel together in fly ash and deposit together on ice. Every study that has used thermal absorption methods to quantify black carbon on glaciers has, without knowing it, been measuring a signal that includes Magnéli phases. How much of the attributed warming belongs to them? Nobody has separated the two contributions, because until very recently nobody knew to try.
Then there’s the biological question, which opens in a different direction.
Yang et al. tested Magnéli phase toxicity in zebrafish embryos and found something unexpected: significant mortality without light exposure. TiO₂ is primarily toxic through photocatalysis — it needs UV light to generate the reactive oxygen species that damage cells. Magnéli phases apparently don’t require it. The mechanism is unknown. The concentrations at which biological effects appear in environmental organisms — not zebrafish embryos in a controlled laboratory, but copepods, pteropods, the small things at the base of the food web that live in water loaded with atmospheric deposition — have not been measured. The fish examined in the microplastics study had Magnéli phases in their gut contents. What that means for those fish, beyond presence, remains an open question.
And then there’s the pattern visible only when the Peru stratigraphy data is laid out in full: the four layers with dramatically elevated light absorption — layers 4, 8, 12, and 16 in the crevasse sequence — fall at perfectly regular intervals. In glacier stratigraphy, regular periodicity usually reflects regular deposition — a recurring atmospheric transport event on a predictable cycle, something that happens on a schedule and leaves a mark in the ice at regular depth. What that schedule is, or what drives it, is not yet known. The pattern is there. The explanation isn’t.
That might be the most honest summary of where this research stands: a gigaton of synthetic mineral distributed across the entire planet, present in the bodies of ocean fish and the walls of Andean glaciers and the sediment of a landlocked Utah lake, and the early chapters of understanding what it does are still being written.
The Sideline, Again
She’d done everything right. Rutile was the correct choice — inert, stable, the same material that goes into food and paper and paint without a second thought. The TiO₂ in that bottle was exactly what it was supposed to be.
The TiO₂ that went into the coal furnace was never meant to be there. It was an accessory mineral, unremarkable, sitting in the rock for 300 million years. Then it went into a furnace at 1,400°C with almost no oxygen, and it came out as something that has never existed in nature — something black, conductive, photocatalytically active, and now present in every glacier and every ocean on Earth.
There’s a coda to that sideline conversation that I couldn’t have offered at the time.
She asked about the photocatalytic forms — anatase, the reactive one. I told her rutile was the safer choice, which is true. And I almost mentioned the one form of titanium oxide that’s genuinely worth paying attention to — not TiO₂ at all, but a family of titanium suboxides produced under extreme heat in low-oxygen environments. Black. Electrically conductive. Photocatalytically active under visible light in ways that TiO₂ is not. The kind of material where you’d know immediately if you were working with it — because nothing in a mineral sunscreen formulation is black.
I didn’t say any of that. I didn’t have a name for it yet. I had a folder.
Now I have a name. And the black ones are already everywhere — in the ice above Huaraz, in the water of the North Pacific, in the sediment of a Utah lake, in the bodies of fish that never came near a coal plant.
The TiO₂ she spread on her skin that afternoon protects her. The TiOx spread across the planet by two centuries of industrial combustion is doing real harm — to glaciers, to ocean chemistry, to organisms whose tolerance for a novel synthetic mineral has never been tested, in concentrations that have never been measured.
She made a good sunscreen. That part of the story is fine.
The other part isn’t.
And here is the difference that will matter long after the rest of this is forgotten.
Radionuclides from nuclear testing, the plastics in ocean sediment, the nitrogen fixed by industrial fertilizers — these are the proposed markers of the Anthropocene, the signals future geologists might use to locate our moment in the rock record. Each has a problem. Radionuclides decay. Plastics degrade over geologic time. Nitrogen cycles out. These are transient signals, legible for thousands or perhaps tens of thousands of years before they dissolve back into background noise. On the timescale of deep geology, they are whispers.
Magnéli phases are structurally analogous to rutile — one of the most geochemically persistent minerals in the rock record, recoverable from sediments hundreds of millions of years old. They are thermally stable, chemically inert, and structurally robust under the same diagenetic conditions that erase most mineralogical evidence of surface processes. They will not dissolve. They will not decay. They will sit in the rock exactly as they were deposited.
And they are everywhere. Not concentrated near industrial centers, not localized to continental margins — but present in high-altitude glacial ice, in the deep floor of the open Pacific, in a landlocked basin in Utah, in the gut contents of fish collected far from any coastline. Synchronous. Global. Diagnostic. A thin layer distributed across the entire planet, deposited within the same narrow window of industrial time.
A geologist reading the rock record half a billion years from now will find that layer. They will recognize it as synthetic — no natural process produces titanium suboxides, and no natural process deposits them simultaneously at every latitude and depth. They will know, from that layer alone, that something happened here: a civilization that burned enough carbon, at high enough temperatures, in low enough oxygen, to transform an accessory mineral that had sat unremarkably in rock for 300 million years into a gigaton of something that had never existed before.
That layer will be us. Permanent, unambiguous, and readable in the dark.
Because the material is black.
The sunscreen she made is white.
Crevasse stratigraphy and LACM absorptive capacity data are from Schmitt et al. (2015), The Cryosphere, 9, 331–340. Magnéli phase discovery and global distribution estimates from Yang et al. (2017), Nature Communications, 8, 194. Raman calibration from Parker & Siegel (1990), Applied Physics Letters, 57, 943. Aragonite dissolution data from Dong et al. (2019), Earth and Planetary Science Letters, 513, 129–141. Wildland-urban interface fire ash as an additional source of Magnéli phases from Baalousha et al. (2026), Environmental Science & Technology, DOI: 10.1021/acs.est.5c09885.
The observations described here span glaciology, oceanography, geobiology, and ichthyology — no single journal owns this pattern, and no single paper could show it whole. That’s why it’s here first. Publicly available and reader accessible.



Fantastic essay, thank you.
So in a nutshell, coal burning has created a new synthetic black mineral impacting global warming and fish in ways we don’t even know yet? The highest to the lowest points?