Everyone Found It Except Us
Gorillas, oncologists, and the U.S. Department of Defense all discovered the same mineral. None of them came through mineralogy.
The Specimen
I bought it for the color.
That’s the honest answer. I’ve been a mineralogist long enough to know better — to walk into a dealer’s room with a list, a budget, a set of analytical questions I’m trying to answer. I know that “pretty” is not a scientific criterion. I know it anyway, and I bought it anyway, because the specimen stopped me. It’s from Colombia, about the size of a dinner plate, and it is extraordinary in the way that only chaotic things can be: hundreds of prismatic quartz crystals radiating outward in every direction, sharp-terminated, a frozen explosion of silica arrested mid-burst. But that’s not what stopped me. What stopped me was the color at the tips. Each termination is stained a warm yellow-orange — not a surface deposit, not something applied after the fact, but halloysite included within the crystal itself during growth, captured as the quartz was forming, like an insect in amber but the other way around: the mineral swallowed by the crystal before either of them finished becoming what they are.
The color is the color of a mango in the last hour before you have to eat it. That particular yellow-orange at the edge of too ripe.



The specimen’s collector name is mango quartz. Its mineralogical name is halloysite-included quartz. But there is a third name — Corona Quartz — which comes from the Corona Mine in Boyacá, Colombia, the only place on Earth where this material has been found. The mine was rediscovered in 2017 after sitting lost in the jungle for nearly two decades, sealed by the man who found it. Specimens began reaching the market in volume around 2019 and 2020. I cannot find a single written record documenting the decision, but the timing is difficult to ignore: the name “Corona” became commercially complicated in early 2020, and somewhere in that same window the trade name shifted to mango quartz. It may be coincidence. I find that I don’t quite believe it is.
When I got it back to the lab I already knew what I was looking at. Halloysite. A clay mineral, common enough in weathered volcanic terrains, in soils above certain basalts, in the oxidized margins of hydrothermal systems. I’ve walked past halloysite specimens a hundred times in the collection. I’ve seen it on labels without stopping.
The mineral is named after Jean-Baptiste Julien d’Omalius d’Halloy — Belgian nobleman, statesman, pioneer of modern geology, born in Liège in 1783. He is the geologist who first defined and named the Cretaceous period, after the chalk strata in the Paris basin.
What I realized, holding the mango quartz under the light, is that I could not have told you what halloysite actually does. I knew the name. I knew the group. I did not know what it was for. What it had been for, across a history far longer than I’d considered.
So I started pulling the thread.
What I found at the other end surprised me enough that I’m still not sure how to say it simply. I’ll try: the same mineral sitting in my specimen case, the same tubular architecture that defines halloysite’s chemistry, has been recognized — independently, by organisms with no shared language and no common laboratory — as one of the most precisely useful structures in the biological world. The gorillas knew. The macaws knew. The oncologists are just catching up.
The Mineral
Halloysite belongs to the kaolin group — the same family as kaolinite, the clay in fine porcelain, in white paper, in the original formulation of antidiarrheal medications that sat in American medicine cabinets for most of the twentieth century. Chemically they are nearly identical: both are aluminum silicate hydroxides, both form in the same weathering environments, both leave the same faint peaks on an X-ray diffraction pattern. A non-specialist looking at the two side by side would struggle to tell them apart.
The difference is geometry.
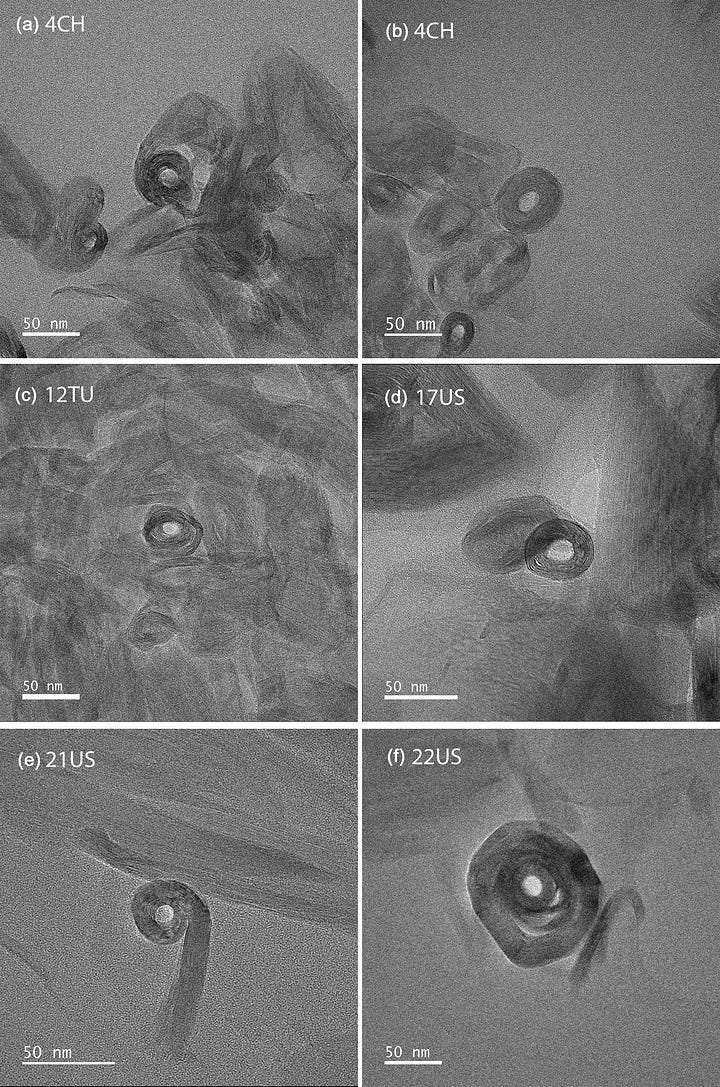
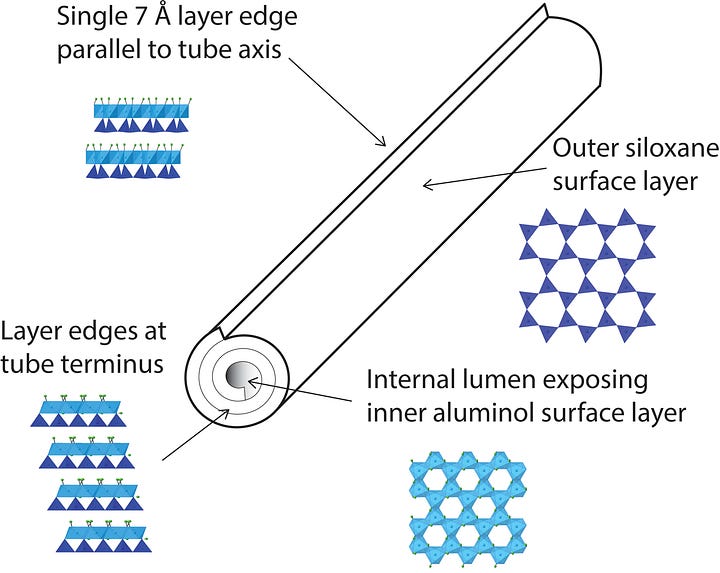
Kaolinite forms flat, hexagonal plates — stacked sheets of linked silicon and aluminum tetrahedra, layered like pages in a book. Halloysite does something stranger. The same sheets, under the right conditions of hydration and surface charge mismatch between the silica and alumina layers, roll. They curl around themselves into hollow cylinders — tubes with an external diameter of roughly 50 to 70 nanometers and an internal diameter of 10 to 20 nanometers, ranging in length from a few hundred nanometers to about a micron and a half. To hold one in your hand, you would need to stack roughly a thousand of them to reach the width of a human hair.
What makes this architecture chemically interesting — what makes it, as it turns out, biologically interesting — is not just the shape but the charge. The inner surface of the tube, lined with aluminum hydroxyl groups, carries a slight positive charge at neutral pH. The outer surface, lined with siloxane groups, carries a slight negative charge. The tube is not simply hollow. It is a hollow cylinder with an inside that behaves differently from its outside. That asymmetry means it can selectively bind different molecules on different surfaces. It means things can be loaded into it, held, and released under the right conditions. It means that, at the nanoscale, halloysite is not just a container. It is a system.
The mango color at the tips of my specimen — the halloysite captured inside the quartz during growth — represents individual tubes too small to resolve without an electron microscope, present in sufficient concentration to tint the crystal like tea steeping in water. What I can see with my eye is the aggregate signal of a geometry I cannot see at all. The color is the evidence. The tubes are the argument.
The Ancient Pole
In the Virunga Mountains of Rwanda, mountain gorillas mine.
The behavior has been documented since Dian Fossey’s fieldwork in the 1960s. Gorillas in the dry season — when their diet shifts toward bamboo, lobelia, and senecio, plants higher in toxic secondary compounds than their usual forage — seek out specific outcrops of weathered volcanic rock. They loosen pieces with their teeth, grind the material to powder with their hands, and eat it. The clay fraction of what they consume has been analyzed. It is dominated by halloysite.
The timing is not coincidental. The shift to more toxic plant foods correlates with increased diarrhea — the body’s mechanism for clearing ingested compounds. The clay consumption follows. What the gorillas appear to be doing, with no laboratory and no periodic table, is adsorbing plant toxins onto the charged surfaces of halloysite nanotubes before those toxins can cross the gut wall. They are, in the functional sense, running a detoxification column in their digestive tract. The mineral is the column packing.

In the tropical forests of Peru, the evidence is more dramatic. On the eroding clay banks of Amazonian rivers, researchers have counted up to 900 parrots from 21 species gathering on a single outcrop — macaws, parakeets, and others arriving before dawn and working the clay face in dense, noisy crowds for hours. In 1999, a research team at UC Davis tested the hypothesis directly. They established that seeds eaten by macaws contain quinidine, a plant alkaloid toxic in sufficient doses. They then fed one group of captive Amazona parrots a mixture of the alkaloid plus clay from a preferred lick, and another group the alkaloid alone. The clay group showed significantly reduced alkaloid absorption — roughly 60% less quinidine in the bloodstream. The birds at the river bank were not eating randomly. The selection was chemically justified.
The clay mineralogy at macaw lick sites is dominated by smectite, with halloysite present in significant quantities. The birds appear capable of distinguishing between outcrops — returning consistently to specific sites even when other clay sources are available nearby. Whether the selection mechanism is taste, texture, or some more cryptic sensory signal is not yet established. That they are selecting is not in question.
These are not isolated cases. Geophagy — the deliberate consumption of earth, clay, or mineral-bearing soil — has been documented across virtually every class of terrestrial vertebrate. Elephants excavate mineral springs. Deer and ungulates seek salt licks with a precision that maps closely onto known mineral deficiencies in local forage. Chimpanzees in West Africa consume specific clay types during periods of intestinal distress. The behavior appears, conservatively, to be as old as the vertebrate gut.
In humans, the record is older than writing. Archaeological evidence of clay consumption appears at sites across sub-Saharan Africa, South America, and Asia. Hippocrates documented it. Avicenna described medicinal clay preparations in the eleventh century. In the American South, kaolin clay consumption — locally called “white dirt” — persisted through the twentieth century as a documented cultural practice, particularly among pregnant women and children. Across sub-Saharan Africa today, studies consistently report geophagy prevalence of between 30 and 65 percent among pregnant women, with consumption of between 100 and 400 grams of clay per day in some regions. The clay is sold in markets. It is mined from specific deposits. It is passed between women as knowledge, generation to generation.
The dominant clay mineral in geophagic soils consumed by pregnant women in Tanzania, Kenya, and Zanzibar has been characterized in multiple independent studies. The answer is consistent: kaolin minerals, frequently halloysite specifically, or a mixture of kaolinite and halloysite. Studies attempting to identify what these materials share — what property might explain why humans and animals converge on them — point repeatedly to the same suite of characteristics: fine particle size, chemical inertness relative to the gut lining, and high adsorption capacity for toxins, pathogens, and plant secondary compounds. The tubular geometry that makes halloysite a candidate for cancer drug delivery is the same geometry that makes it an effective gut-lining buffer. The mechanism is the same. The application is separated by a hundred million years of evolutionary trial and a few decades of materials science.
What the clinical literature calls pica — the consumption of non-food substances, classified as a disorder when it presents in a Western medical context — is, when examined mineralogically, a behavior with a chemically coherent rationale that spans the entire vertebrate lineage. The gorilla grinding clay in the dry season is not exhibiting aberrant behavior. It is running a protocol that works.
The Bridge
The mineral doing all of this is halloysite.
The same tubular geometry that a mountain gorilla selected empirically in the Virunga Mountains, that pregnant women across four continents have been mining from specific deposits and passing between generations as knowledge — that geometry is now the subject of several hundred peer-reviewed papers in materials science and nanomedicine. For most of the twentieth century, a version of this clay sat quietly in American medicine cabinets under a brand name that didn’t mention minerals at all. Nobody connected the dots. The discipline that owns the object wasn’t looking.
The Frontier
The first thing materials scientists discovered about halloysite was the same property the gorillas had been exploiting for centuries: the tube.
The hollow lumen of a halloysite nanotube — 10 to 20 nanometers in internal diameter, up to a micron and a half in length — turns out to be nearly ideal for loading and releasing chemical compounds in a controlled way. The inner aluminol surface holds a slight positive charge. The outer siloxane surface holds a slight negative charge. Those opposing charges mean the tube can be loaded with different molecules on different surfaces simultaneously — one compound inside the lumen, another bound to the exterior — and the two payloads can be engineered to release under different conditions. pH. Temperature. Enzymatic activity. The tube doesn’t just carry. It responds.
In cancer drug delivery, this matters enormously. One of the persistent problems in chemotherapy is that cytotoxic drugs don’t distinguish well between tumor tissue and healthy tissue — the drug goes everywhere, and the damage follows. Halloysite nanotubes offer a partial solution. Loaded with doxorubicin, one of the most widely used chemotherapy agents, halloysite nanotubes release their payload preferentially in acidic environments. Tumor tissue is acidic. The pH-triggered release means the drug concentrates where it’s needed and spares, to some degree, the tissue around it. Studies have demonstrated this effect in cervical adenocarcinoma cells, breast cancer cells, and leukemia cell lines. The nanotube has been decorated with folic acid to actively target cancer cells that overexpress the folate receptor. It has been conjugated with magnetic particles for guided delivery. It has been used to carry two different anticancer agents simultaneously — one inside the lumen, one on the exterior — with independent release profiles.
Halloysite nanotubes have also been shown to cross the blood-brain barrier, one of the most formidable obstacles in neuropharmacology. A sustained delivery system that can carry a drug payload across that barrier and release it gradually, without killing the cells it’s trying to treat, is not a minor achievement. The clay is doing this. The clay that forms in weathered volcanic rock, that a gorilla grinds with its hands in the dry season, that a pregnant woman in Zanzibar selects from a specific deposit and carries home.
The mechanism in every case is the same one Act 2 described: the tube, the charge asymmetry, the selective surface chemistry. What the researchers are engineering with functionalized polymers and pH-responsive bonds is a refined version of what the halloysite nanotube does naturally. The sophistication is real. But the architecture was already there, in the mineral, waiting to be described.
There are now several hundred peer-reviewed papers on halloysite nanotube biomedical applications. The literature is almost entirely in materials science, chemistry, and nanomedicine journals. Search “halloysite” in a mineralogy database and the results thin considerably. The object is the same. The disciplines looking at it are not.
The Gap
Here is a simple test.
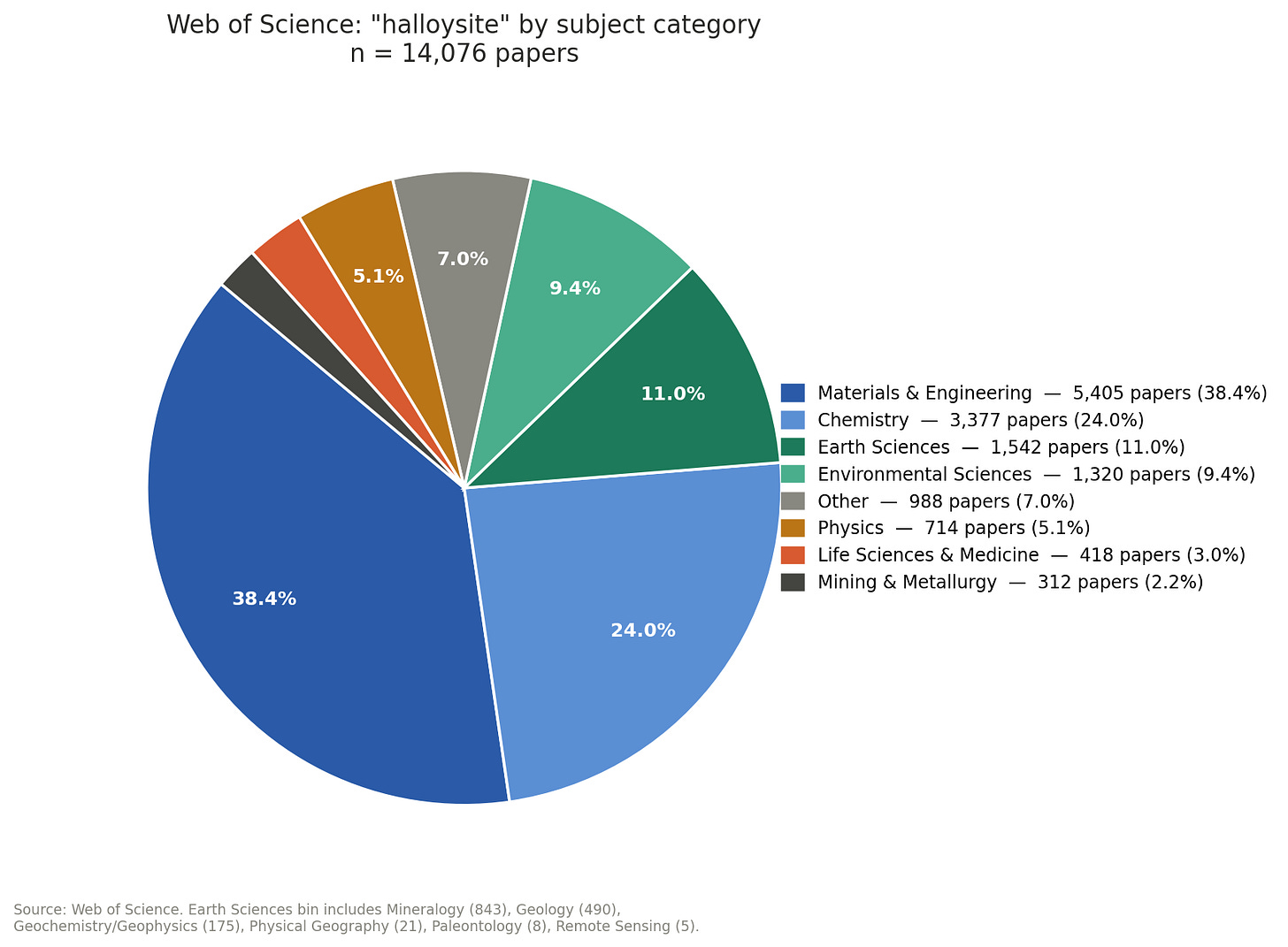
Search “halloysite” in Web of Science. As of this writing, the database returns results across more than fifty subject categories. Chemistry leads with 3,193 papers. Materials Science follows with 2,870. Engineering contributes 1,291. Polymer Science adds 1,174. Combined, those four applied science categories account for roughly 9,500 papers about a mineral.
Mineralogy appears sixth on the list, with 843 papers. Geology adds 490. Together, the two disciplines that own the object — that named it, characterized its crystal structure, and maintain the reference collections — account for roughly 11% of all papers written about it. That figure is itself generous: the Earth Sciences bin includes structural crystallography, geological occurrence studies, and XRD characterization work — papers describing what halloysite is, not what it does. The literature engaging with halloysite’s biological interactions, its behavior in living systems, its potential in medicine, sits almost entirely outside those 1,333 papers. Notably, Oncology as a subject category returns just 2 papers — the cancer drug delivery research described in the previous section is being published and classified under Chemistry and Materials Science. The clinical literature hasn’t caught up yet, which means the gap may look different in a decade. But right now, the mineral is doing oncology work that oncology journals have barely begun to claim.
The geophagy literature barely registers. Nutrition and Dietetics: 20 papers. Zoology: 5. Veterinary Sciences: 6. The biological behavior that motivated the mineral’s selection across the entire vertebrate lineage, independently, across millions of years — the behavior that is, as this post has argued, the first evidence of halloysite’s functional chemistry — generates less than 0.3% of the total literature.
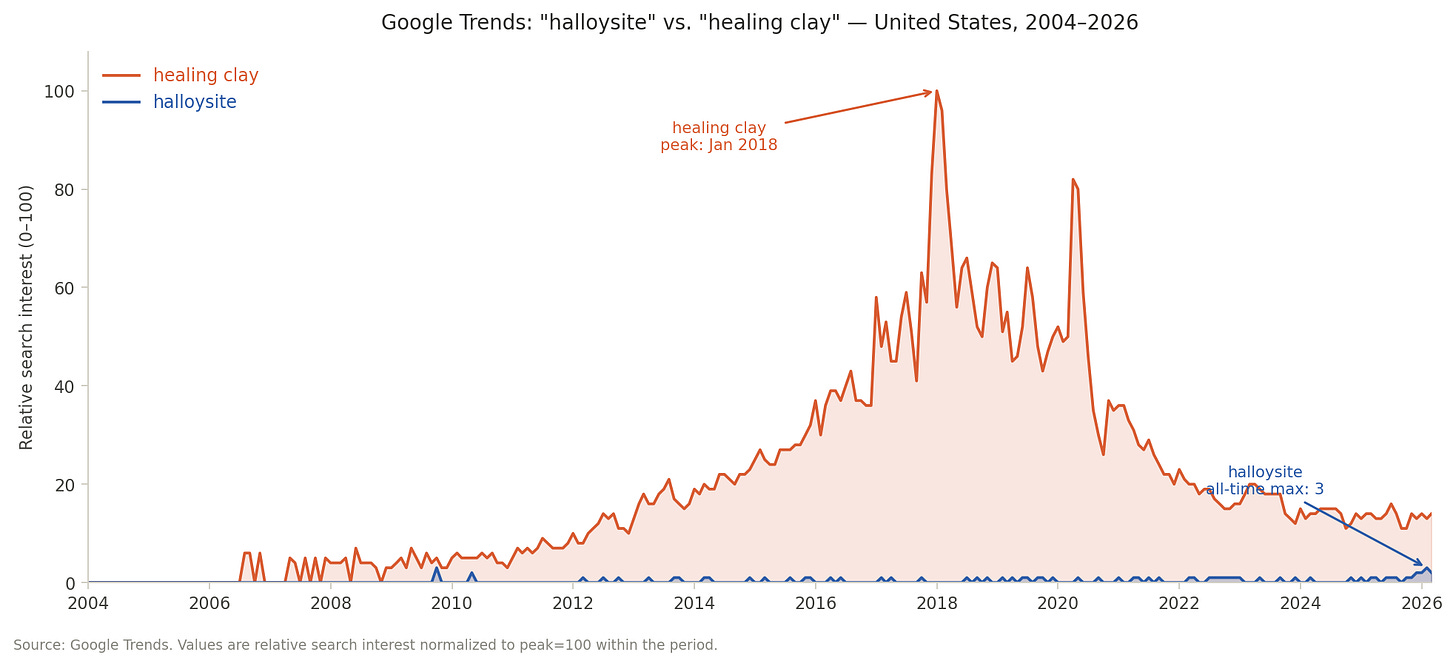
Run a parallel search on Google Trends. Over 22 years of US search history, “halloysite” has never exceeded a relative search index of 3 — its all-time maximum, reached twice, in October 2009 and February 2026. “Healing clay” peaked at 100 in January 2018 and has averaged 22 since 2020, while halloysite has averaged 0.5 over the same period. That is a ratio of roughly 45 to one. The public searching for the mineral’s biological properties — the gut chemistry, the adsorption behavior, the thing this post is about — is not finding mineralogy. It is finding wellness culture. The discipline ceded the vocabulary and never reclaimed it.
The audience exists. The animals worked out the mechanism. The oncologists are publishing the results. And as of late 2025, the U.S. Department of Defense has a fourth reason to care. In December 2025, a Utah-based company confirmed a major halloysite deposit in the Lake Mountains and announced it is deriving nano-silicon directly from the nanotube structure — the same hollow cylinder described in this post — to produce EV battery anodes that charge to 80% capacity in under five minutes, compared to 40 minutes or more for conventional graphite batteries. The tube is the reason that works. On February 2, 2026, the U.S. government announced Project Vault, a $10 billion strategic reserve for critical minerals, with halloysite-hosted deposits in the conversation. A preliminary economic assessment is still pending, and the commercial claims deserve scrutiny before a feasibility study validates them. But the geometry that a gorilla selected empirically in Rwanda is now the subject of a federal industrial policy announcement.
Mineralogy — the discipline that named this mineral, that characterized its structure, that maintains the reference collections — is present in the literature, but peripheral to all four conversations it should be leading. The oncologists, the materials scientists, the wellness community, and now the defense supply chain strategists have all arrived at halloysite independently. None of them came through mineralogy to get there.
The Close
The mango quartz is still on my desk.
I keep moving it to make room for other things and then moving it back. I tell myself it’s because it’s beautiful, which is true. But I think the real reason is that I’m not done looking at it yet. Not because I don’t know what it is — I know considerably more than I did when I bought it — but because knowing what it is has not made it smaller. It has made it stranger.
The color at the tips is still the color of a mango in the last hour before you have to eat it. The halloysite is still there inside the quartz, invisible at any scale I can access without an electron microscope, present only as aggregate signal — the warm yellow-orange that stopped me at the dealer’s table. The tubes are still there too, doing what they have always done: holding geometry that living systems have been finding useful for longer than our genus has existed.
A gorilla in the dry season found it useful. A macaw at a river bank found it useful. A pregnant woman in Zanzibar, selecting clay from a specific deposit and carrying it home, found it useful. A materials scientist loading doxorubicin into a nanotube for targeted delivery to a breast cancer cell is finding it useful right now, in a lab somewhere, having arrived at the same architecture by a completely different road.
The mineral didn’t change. It was always this.
I bought it for the color. I kept it for everything else.
References
Fossey, D. (1983). Gorillas in the Mist. Houghton Mifflin.
Gilardi, J. D., Duffey, S. S., Munn, C. A., & Tell, L. A. (1999). “Biochemical functions of geophagy in parrots: Detoxification of dietary toxins and cytoprotective effects.” Journal of Chemical Ecology, 25(4), 897–922. https://doi.org/10.1023/A:1020857120217
Krishnamani, R., & Mahaney, W. C. (2000). “Geophagy among primates: Adaptive significance and ecological consequences.” Animal Behaviour.
Mahaney, W. C., et al. (1990). “Geophagia by mountain gorillas (Gorilla gorilla beringei) in the Virunga Mountains, Rwanda.” Primates.
Mahaney, W. C., et al. (1995). “Mountain gorilla geophagy: A possible seasonal behavior for dealing with the effects of dietary changes.” International Journal of Primatology.
Young, S. L., Wilson, M. J., Hillier, S., Delbos, E., Ali, S. M., & Stoltzfus, R. J. (2010). “Differences and commonalities in physical, chemical and mineralogical properties of Zanzibari geophagic soils.” Journal of Chemical Ecology, 36(1), 129–140. https://doi.org/10.1007/s10886-009-9729-y
Young, S. L., & Miller, J. D. (2019). “Medicine beneath your feet: A biocultural examination of the risks and benefits of geophagy.” Clays and Clay Minerals, 67(1), 81–90. https://doi.org/10.1007/s42860-018-0004-6
Gray-Wannell, N., Cubillas, P., Aslam, Z., Holliman, P. J., Greenwell, H. C., Brydson, R., Delbos, E., Strachan, L.-J., Fuller, M., & Hillier, S. (2023). "Morphological features of halloysite nanotubes as revealed by various microscopies." Clay Minerals, 58, 395–407. https://doi.org/10.1180/clm.2023.37 — SEM, AFM, and cross-sectional TEM study of nine halloysite samples; establishes mean lumen diameter of 12 nm and demonstrates that the standard "carpet roll" model is an oversimplification. Open access.
Davies, T. C. (2023). “Current status of research and gaps in knowledge of geophagic practices in Africa.” Frontiers in Nutrition, 9. https://doi.org/10.3389/fnut.2022.1084589
Macaw photo: https://commons.wikimedia.org/wiki/File:Parrots_at_a_clay_lick_-Tambopata_National_Reserve,_Peru-8c.jpg


Another excellent article Aaron. I think I'll look at Mango Quartz differently after reading all that. But I still won't be chasing one for my collection. Too much marketing hype 😁